Pfizer’s arthritis drug Xeljanz® has been linked to an increased risk of heart attacks, strokes, cancer, blood clots and death.
What You Can Do & How We Can Help
The Schmidt Firm, PLLC is currently accepting Xeljanz induced injury cases in all 50 states. If you or somebody you know has been diagnosed with cancer, a major cardiovascular event, blood clots, or death after taking Xeljanz, you should contact our lawyers immediately for a free case consultation. Please use the form below to contact our Defective Drug Litigation Group or call toll free 24 hours a day at (866) 920-0753.
FDA Orders “Black Box” Warning for Xeljanz Side Effects
In September 2021, the FDA required “Black Box” warnings about deadly side effects of Xeljanz, including:
- Heart-related events
- Heart attacks
- Strokes
- Cancer (particularly lymphoma and lung cancer)
- Blood clots
- Death
What is Xeljanz?
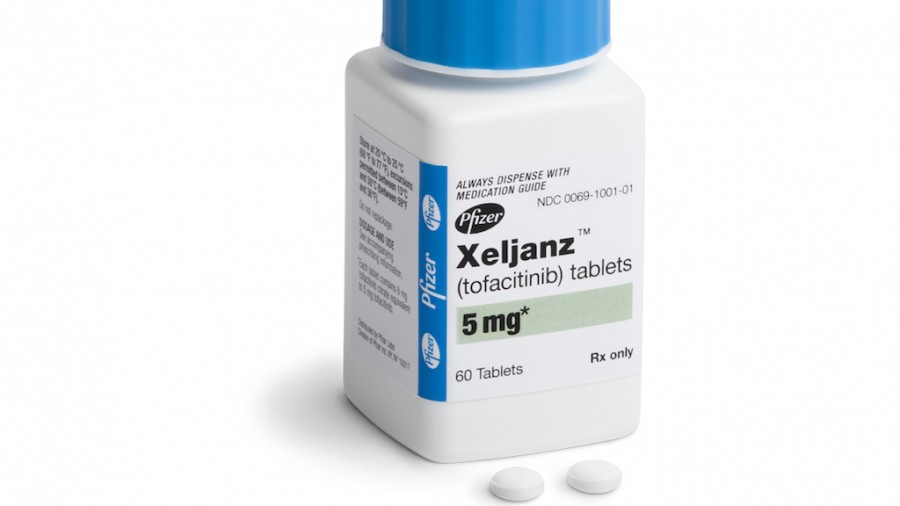
Xeljanz and Xeljanz XR (generic: tofacitinib) are prescription medications that are manufactured by Pfizer. Xeljanz was approved for the treatment of adults with moderately to severely active rheumatoid arthritis in November 2012.
Since then, Xeljanz has also been approved to treat psoriatic arthritis (2017), ulcerative colitis (2019), and juvenile idiopathic arthritis (2020). It is a blockbuster drug for Pfizer, bringing in over $2 billion in revenue in both 2019 and 2020.
What are JAK Inhibitors?
Xeljanz is the #1 most-prescribed drug in a class of anti-inflammatory medications called Janus Kinase Inhibitors (JAK inhibitors). These medications are immunosuppressant drugs, which means they decrease the activity of the immune system.
How Do They Work?
Xeljanz and other JAK inhibitors work by blocking the activity of cytokines, which are proteins that fight off infections by causing inflammation in the body. Other JAK inhibitors include Olumiant® (baricitinib) and Rinvoq® (upadacitinib).
Xeljanz and Cancer
Xeljanz is linked to an increased risk of all types of cancer (except non-melanoma skin cancer), but it is strongly associated with lymphomas and lung cancer in people who are current or former smokers.
Xeljanz and Major Cardiac Events
Some of the most serious heart-related events linked to Xeljanz include:
- Cardiac arrest
- Heart attack
- Ischemic stroke
- Acute coronary syndrome
- Atrial fibrillation
- Heart failure
- Death
Xeljanz and Blood Clots
Xeljanz is linked to an increased risk of blood clots, which can develop in the legs and travel to the lungs, resulting in severe injury or death. Blood clots in the legs are also called Deep Vein Thrombosis (DVT). If a blood clot gets trapped in the lungs, it is known as a Pulmonary Embolism or Arterial Thrombosis.
Pfizer Study Links Xeljanz With Deadly Risks
In February 2021, the FDA reported that Xeljanz was associated with serious heart-related problems and cancer. The warnings were based off a study that Pfizer was required to complete after Xeljanz was approved in 2012. The study compared the safety of Xeljanz at two doses (5-mg twice daily and 10-mg twice daily) with another type of arthritis drug called a TNF inhibitor.
Black Box Warning for Blood Clots & Death
In July 2019, the FDA added a Boxed Warning on Xeljanz regarding the increased risk of blood clots and death from the 10-mg twice-daily dose. Patients should stop taking Xeljanz and seek emergency medical attention if they develop symptoms of a blood clot, such as:
- Sudden shortness of breath
- Chest pain that worsens with breathing
- Swelling of a leg or arm
- Leg pain or tenderness, or red or discolored skin in the painful or swollen leg or arm
Xeljanz Linked to Blood Clots and Death
In February 2019, the FDA issued a Safety Announcement to warn that 10-mg twice-daily doses of Xeljanz increases the risk for death and life-threatening blood clots in the lungs of patients with rheumatoid arthritis.
“A safety clinical trial found an increased risk of blood clots in the lungs and death when a 10-mg twice-daily dose of tofacitinib (Xeljanz, Xeljanz XR) was used in patients with rheumatoid arthritis (RA).”
The FDA has not approved this 10-mg twice-daily dose for the treatment of rheumatoid arthritis. This dose is only approved in the dosing regimen for patients with ulcerative colitis.
Xeljanz and Rheumatoid Arthritis
Xeljanz® (tofactinib) was approved by the FDA for the treatment of rheumatoid arthritis in 2012. It works by decreasing the activity of the immune system. In patients with rheumatoid arthritis, the immune system attacks its own joints, causing severe pain and swelling.
Xeljanz Safety Study
As a condition of approval, the FDA required Pfizer to conduct a study to investigate the risk of possible side effects like heart-related events, cancer, and certain infections.The study involved patients who were at least 50 years old with at least one cardiovascular risk-factor.
Safety Committee Finds Higher Risk for Pulmonary Embolism and Death
During the analysis of the study, an external safety-monitoring committee found an increased risk of death and blood clots in the lungs (pulmonary embolism) in patients who were taking Xeljanz (10-mg) twice-daily compared to to patients who were taking Xeljanz (5-mg) twice-daily or a TNF inhibitor.
FDA: Doctors Should Prescribe Recommended Doses of Xeljanz
The FDA urges doctors to prescribe Xeljanz at the recommended doses. The 10-mg twice-daily dose that is linked to blood clots and death is NOT approved for rheumatoid arthritis patients — however, it is approved for patients with ulcerative colitis.
What Should I Do?
Doctors should also watch patients for signs and symptoms of blood clots or pulmonary embolism. Patients should be instructed to seek medical care immediately if these symptoms occur, the FDA warned.
Warning Signs & Symptoms of Pulmonary Embolism
- Sudden trouble breathing
- Shortness of breath
- Chest pain (or back pain)
- Coughing up blood
- Severe sweating
- Skin that is clammy or bluish-colored
Do I have a Xeljanz Lawsuit?
The Schmidt Firm, PLLC is currently accepting Xeljanz induced injury cases in all 50 states. If you or somebody you know has suffered from cancer, a heart attack, stroke, blood clots, or death after taking Xeljanz, you should contact our lawyers immediately for a free case consultation. Please use the form below to contact our Defective Drug Litigation Group or call toll free 24 hours a day at (866) 920-0753.
Attention Lawyers: We consider a referral from another law firm to be one of the greatest compliments. If your firm is interested in referring us a case or for us to send you a list of previous award judgments and/or average referral fees, please visit the Lawyer Referral section of our website.

 The Schmidt Firm, PLLC has been recognized as one of the nation’s leading plaintiffs' law firms and handles cases in all 50 states. We are very proud of our legal achievements, but equally self-respecting of our firm's reputation for providing personal attention to each and every client we represent.
The Schmidt Firm, PLLC has been recognized as one of the nation’s leading plaintiffs' law firms and handles cases in all 50 states. We are very proud of our legal achievements, but equally self-respecting of our firm's reputation for providing personal attention to each and every client we represent.

