The Cordis OptEase is meant to prevent deadly pulmonary embolisms, but it has been linked to life-threatening risks like fracture, migration, organ damage, and more.
What You Can Do & How We Can Help
The Schmidt Firm, PLLC is currently evaluating IVC filter cases in all 50 states, regardless of whether you have been injured or not. If you or somebody you know was implanted with an IVC filter, you should contact our lawyers immediately for a free case consultation. Please use the form below to contact our Defective Medical Device Litigation Group or call toll free 24 hours a day at (866) 920-0753.
Overview
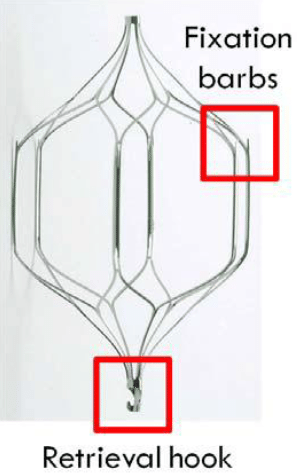
Cordis Corporation manufactures the OptEase inferior vena cava (IVC) filter. It is a temporary implant designed to be removed when it is no longer necessary. The longer it remains implanted, the higher the risk of complications.
Design Based on Controversial Filters
A big problem is that its design is closely related to the C.R. Bard Recovery filter, which was withdrawn from the market in 2005 after being linked to a high risk of fracture. However, because the OptEase was approved with a 510(k) application, it has never been studied for safety in clinical trials and the long-term risks are unknown.
OptEase Recall
In March 2013, about 33,000 OptEase filters were recalled because a labeling error might cause it to be implanted backward. If this occurred, there would be nothing to stop the filter from migrating in the bloodstream to a patient’s heart. Click here to read more.
FDA Safety Recommendations
Due to increased risks like filter fracture, migration, and organ damage, the FDA recommends removing IVC filers 29-54 days after implantation. However, studies have found that real-world retrieval rates remain as low as 20%.
The FDA has issued a Safety Communication to warn about side effects, such as:
- Filter fracture
- Migration or tilting
- Embolization of broken pieces of the filter to other parts of the body
- Perforation through the vena cava
- Organ damage (heart, lungs, kidneys, etc.)
- Internal bleeding
- Heart problems
- Doctors may be unable to retrieve broken filter
- And more
IVC Filter Lawsuits Centralized in Multi-District Litigation (MDL)
Hundreds of people who were seriously injured by side effects of IVC filters have filed lawsuits. Judges have centralized cases against C.R. Bard and Cook Medical in federal courts in Arizona and Indiana. However, this litigation is not a class action — they are individual lawsuits in a Multi-District Litigation (MDL) and each case can have its own outcome.
What is the problem?
The Schmidt Firm, PLLC is nationally recognized as a class action law firm. However, our lawyers are filing individual lawsuits against Cordis Corporation rather than a a class action lawsuit. If you were injured by the OptEase, you could be entitled to compensation.
About Class Actions
There are many advantages to a class action when a lot of people have similar legal claims. Everyone joins together to collectively seek compensation. Any payout is divided and shared equally.
Why Our Law Firm is Filing Individual Lawsuits as Opposed to a Class Action
Class actions are designed for efficiency — not maximizing compensation for individuals with severe injuries from defective medical devices. Members may have to accept “low-ball” settlements or massive attorneys’ fees that leave plaintiffs little to share.
Our attorneys file individual lawsuits so we can focus our efforts on helping people with the most serious injuries. We specialize in seeking justice and maximize compensation from device-makers who fail to warn about side effects. If you were injured by an IVC filter, we may be able to help you seek compensation for you pain and suffering, medical expenses, lost income, and more.
Do I have an IVC Filter Lawsuit?
The Schmidt Firm, PLLC is evaluating IVC filter cases in all 50 states, regardless of whether you were injured or not. If you or someone you know received an IVC filter implant, please contact our lawyers immediately for a free case consultation. You may be entitled to compensation by filing a lawsuit.
Please use the form below to contact our Defective Medical Device Litigation Group or call toll free 24 hours a day at (866) 920-0753.
Attention Lawyers: We consider a referral from another law firm to be one of the greatest compliments. If your firm is interested in referring us a case or for us to send you a list of previous award judgments and/or average referral fees, please visit the Lawyer Referral section of our website.

 The Schmidt Firm, PLLC has been recognized as one of the nation’s leading plaintiffs' law firms and handles cases in all 50 states. We are very proud of our legal achievements, but equally self-respecting of our firm's reputation for providing personal attention to each and every client we represent.
The Schmidt Firm, PLLC has been recognized as one of the nation’s leading plaintiffs' law firms and handles cases in all 50 states. We are very proud of our legal achievements, but equally self-respecting of our firm's reputation for providing personal attention to each and every client we represent.

